University Health Network (UHN) is a research hospital affiliated with the University of Toronto and a member of the Toronto Academic Health Science Network. The scope of research and complexity of cases at UHN have made it a national and international source for discovery, education and patient care.
Recent News

Meet Dr. Jennifer Jones @PMResearch
From surviving to thriving: How research is reshaping life after cancer
Read MoreMeet Dr. Jennifer Jones @PMResearch
From surviving to thriving: How research is reshaping life after cancer

Image Caption: Dr. Jennifer Jones is a Senior Scientist, Director of the Cancer Rehabilitation and Survivorship Program, and the Butterfield/Drew Chair in Cancer Survivorship Research at UHN’s Princess Margaret Cancer Centre.
More people are surviving cancer today than ever before, due to recent advancements in early detection and treatments.
However, survival is only part of the story. Cancer and its treatments often leave people with long-term effects such as fatigue, pain, weakness, memory problems, and emotional distress. These challenges can last months or years, making it hard to return to work, care for family, and enjoy everyday life.
“Cancer changes everything,” says Dr. Jennifer Jones, Senior Scientist and Director of the Cancer Rehabilitation and Survivorship Program at UHN’s Princess Margaret Cancer Centre (PM). “For many patients, the challenge isn’t just getting through treatment; it’s what comes after.”
A new model of care: Transforming life after cancer
Jennifer leads the Cancer Rehabilitation and Survivorship (CRS) Program at PM, which is one of the largest comprehensive programs of its kind worldwide.
Patients with cancer-related impairments and disability are referred to CRS to receive a full assessment, a personalized plan to address their needs, and sometimes 1:1 follow-up consults.
Patients who have several impairments and deconditioning are typically enrolled in the CaRE program, which supports patients with eight weeks of tailored progressive exercise training and self-management classes addressing common side effects such as fatigue, cognitive impairments, and dealing with difficult emotions.
The CaRE program can be delivered in person by a multidisciplinary CRS team in small groups at the ELLICSR Centre for Health, Wellness and Cancer Survivorship, which was funded by the Canadian Foundation for Innovation. The CaRE@ELLICSR program also places a focus on nutrition, providing hands-on cooking classes with a wellness chef and dietitian. A prospective study of CaRE@ELLICSR showed that it improved patients’ physical activity, upper body muscular strength, and cancer-related symptoms.
“We see people regain strength, confidence, and control over their lives,” she says. “That’s what drives this work.”

CaRE@ELLICSR provides multi-dimensional rehabilitation care in group settings. Patients receive tailored exercise prescriptions, nutrition, and self-management classes to address their needs.
Research in a dynamic real-life situation
What sets the CRS program apart is how tightly care and research are connected.
Rather than treating research and clinical care as separate, Jennifer and her team have built an integrated model where each informs the other. Jennifer learned the model from her mentors, Dr. Gary Rodin, who founded the Department of Supportive Care at Princess Margaret, and Dr. Pamela Catton, former Director of Oncology Education and Cancer Survivorship at UHN.
When Dr. Catton passed away, Jennifer assumed the role of Director of the Cancer Rehabilitation and Survivorship Program. She carried on her legacy and their collective research that paved the way for the current program.
“What we study comes directly from what we see in the clinic,” she explains. “We identify the gaps, test solutions, and then bring those solutions right back into care.”
This approach has led to the rapid development and testing of new models like CaRE@Home, a virtual program designed to improve access. Early studies showed it is feasible, safe, and helps reduce disability. It is now being evaluated in a multi-centre Phase III trial funded through the Canadian Institutes of Health Research (CIHR) and the Canadian Cancer Society.
Other models of the CaRE program have been developed based on different patient needs: CaRE-Advanced Cancer, which was developed for people living with metastatic disease, helps patients maintain function, manage ongoing treatment effects, and sustain life roles. And CaRE-AYA was launched to address the specific needs of adolescents and young adults under 40.
Tailoring care for those who need it most
The same model is driving innovation for patients with more complex needs.
In 2019, Dr. Jonas Mattsson, Director of the Hans Messner Allogeneic Transplant Program at PM, contacted Jennifer to see how they could help patients undergoing allogeneic blood and marrow transplantation (allo-BMT) to be more active, so that they can better withstand and recover from transplantation.
Together, the two teams developed CaRE-4-AlloBMT, which is a longitudinal rehab program delivered before transplant, during hospitalization, and then 6 months after transplant.
“AlloBMT patients face many physical challenges from previous cancer treatments and the transplant itself. Incorporating multidimensional rehabilitation from pre- to post-transplantation is the ideal clinical approach. This can provide both a preventive ‘buffering’ and a restorative supportive measure,” says Jennifer.
In a Phase II trial, CaRE-4-AlloBMT was found to be feasible, acceptable, and safe, with promising results related to improving physical function and disability. To build on this, the team is now conducting a larger Phase III trial funded through CIHR.
Jennifer is excited and optimistic about this study: “This is a proactive approach that can bring promising benefits to preserve and optimize physical function, address alloBMT-related morbidities, minimize dysfunction, and enhance quality of life.”
Changing the future of cancer recovery
For Jennifer, the ultimate goal extends beyond a single program.
“We have a responsibility to build the evidence around cancer rehabilitation,” she says. “If we can show these programs work, we can make the case for broader access so patients across Canada can receive the care they need.”
Through an integrated model of clinical care and research, her team is not only helping patients recover, but they are redefining what recovery after cancer can look like.
Reflecting on the important things in life
Through her work with patients, Dr. Jennifer Jones has gained a unique perspective on what truly matters in life.
She has seen how a cancer diagnosis reshapes priorities, shifting focus away from work, status, and material things, and toward family, relationships, and meaningful experiences.
“People often come out of cancer with a very different lens,” she says. “They think more about how they want to spend their time and who they want to spend it with.”
That perspective has shaped how she approaches her own life.
“I work hard, but when I leave work, I’m fully present with my family,” she says. “That balance is incredibly important to me.”
She and her family also try to live the same healthy, active lifestyle she encourages in her patients, something she sees not as an obligation, but as an investment in long-term well-being.
“Don’t wait for a health crisis to start making changes,” she says. “Small, consistent steps, staying active, prioritizing your mental health, and making time for the people in your life, can make a huge difference.”
It is a message that reflects both her research and her lived experience:
“More people are surviving cancer. The goal now is to live well, and that’s something we should all be thinking about.”
Meet PMResearch is a story series that features Princess Margaret researchers. It showcases the research of world-class scientists, as well as their passions and interests in career and life—from hobbies and avocations to career trajectories and life philosophies. The researchers that we select are relevant to advocacy/awareness initiatives or have recently received awards or published papers. We are also showcasing the diversity of our staff in keeping with UHN themes and priorities.
×

How the Body Remembers Inflammation
Researchers discover how repeated infections and stress can change blood stem cells over time.
Read MoreHow the Body Remembers Inflammation
Researchers discover how repeated infections and stress can change blood stem cells over time.

Image Caption: Newly identified blood stem cells, called HSC inflammatory memory (HSC-iM) cells, are distinct from other blood stem cells in that they develop changes at the molecular level in response to inflammation that are maintained long term.
Stress from inflammation over long periods of time can drive the aging of blood cells and raise the risk of cancer. In a new study published in Nature, researchers at UHN’s Princess Margaret Cancer Centre (PM) and the University of Oxford have identified human blood stem cells (hematopoietic stem cells, HSCs) that can remember prior inflammation—a finding that could impact what is known about aging and the long-term effects of inflammation.
Mature blood cells are continuously replenished by bone marrow hematopoietic stem and progenitor cells—rare, self-renewing cells that develop into all blood cell types. Inflammation, caused by infections and other stressors over time, can contribute to an age-related decline in HSCs.
It is not fully known how daily blood production is coordinated with the maintenance of blood stem cells over a lifespan, especially in response to repeated episodes of inflammation. To understand how HSCs adapt and respond to inflammation, the research team, led by senior authors Drs. Stephanie Xie and John Dick from PM and Dr. Paresh Vyas from the MRC Weatherall Institute of Molecular Medicine at the University of Oxford, generated specific lab models of inflammation to analyze human HSCs.
These models, called xenograft inflammation-recovery models, use human cells to study inflammation by exposing them to conditions that mimic inflammatory stress, such as those seen in sepsis or aging. These models are then analysed at the molecular and single-cell level.
The research team identified a distinct group of HSCs that differed at the level of how their genes work. These cells were termed HSC inflammatory memory cells (HSC-iM) as they could retain the memory of past inflammation. After exposure to inflammatory treatments, HSC-iM retained molecular changes that included long-lasting shifts in how the stem cells behaved and communicated. They also remained in a resting state and produced fewer blood cells than usual.
Indications of this inflammatory memory were also found in HSC samples from people recovering from COVID-19, individuals with sickle cell disease, older adults, and people with age-related blood cell changes—validating the results from the lab models.
Interestingly, in situations where HSCs were impacted by a condition caused by age-related genetic mutations, called clonal hematopoiesis, the impact of inflammatory stress was reduced. HSC-iM with these changes were more likely to be activated and divide to mature into working blood cells. In other words, these mutations made the memory cells behave differently.
The molecular differences observed in HSC-iM were also passed down to immune cells that these stem cells matured into, shaping how those cells behave and react to inflammation later on.
Additionally, people whose circulating blood cells showed more of these long-lasting inflammatory patterns had a higher overall risk of death, linking these stem cell changes to real-world health outcomes and helping explain why inflammation affects people differently across a lifetime.
“These findings help explain why inflammatory experiences earlier in life may shape a person’s health decades later,” says Dr. Stephanie Xie, Scientist at UHN’s Princess Margaret Cancer Centre and corresponding author of the study. “It provides us with more tools to investigate how health outcomes due to aging and age-associated diseases can differ between individuals.”
Dr. Andy Zeng is the co-first author of the study and an MD/PhD candidate at UHN and the Temerty Faculty of Medicine at the University of Toronto.
Dr. Murtaza Nagree is the co-first author of the study and a Postdoctoral Researcher at UHN.
Dr. Niels Asger Jakobsen is the co-first author of the study and a Postdoctoral Research Scientist at the University of Oxford.
Dr. Paresh Vyas is the co-senior author of the study. He is a Professor of Haematology at the University of Oxford.
Dr. John Dick is the co-senior author of the study. He is a Senior Scientist at UHN’s Princess Margaret Cancer Centre, the Helga and Antonio De Gasperis Chair in Blood Cancer Stem Cell Research, a Professor in the Department of Molecular Genetics at the Temerty Faculty of Medicine at the University of Toronto, and University Professor, University of Toronto.
Dr. Stephanie Xie is the co-senior and corresponding author of the study. She is a Scientist at UHN’s Princess Margaret Cancer Centre and an Assistant Professor in the Department of Medical Biophysics at the Temerty Faculty of Medicine at University of Toronto.
This work was supported by the University of Toronto, the Medical Research Council, Leukaemia UK, Blood Cancer UK, the Government of Ontario, the International Development Research Centre, the Government of Canada, the Terry Fox Research Institute, Blood Cancer United, the Canadian Institutes for Health Research, the Canadian Cancer Society, the Medical Research Council (MRC) Molecular Haematology Unit at the University of Oxford, the NIHR Oxford Biomedical Research Centre, the Allan Slaight Breakthrough Fund, and The Princess Margaret Cancer Foundation.
Dr. John Dick receives royalties from Trillium Therapeutics Inc./Pfizer.
Zeng AGX, Nagree MS, Jakobsen NA, Shah S, Varesi A, Kang JRW, Murison A, Cheong JG, Turkalj S, Zhang X, Radtke FA, Abera TA, Lim INX, Jin L, Araújo J, Aguilar-Navarro AG, Parris D, McLeod J, Kim H, Lee HS, Zhang L, Boulanger M, Bader E, Gbeha E, Parkhurst CN, Wagenblast E, Flores-Figueroa E, Wang B, Schwartz GW, Shultz LD, Nam AS, Grimes HL, Josefowicz SZ, Awadalla P, Vyas P, Dick JE, Xie SZ. Human haematopoietic stem cells remember inflammatory stress. Nature. 2026 May 27; doi:10.1038/s41586-026-10522-7.
×

The Art of Transplant Experience
Exploring lived experience through art reveals deeper insights into organ transplantation.
Read MoreThe Art of Transplant Experience
Exploring lived experience through art reveals deeper insights into organ transplantation.

Image Caption: The artwork created by participants illustrates how arts-based approaches can capture the emotional and lived experiences of organ transplantation. (Image courtesy of Dr. Suze Berkhout)
Organ transplantation is often described as a life-saving intervention focused on positive clinical outcomes and recovery. At the same time, lived experiences are complex and include emotional challenges and mixed feelings about treatment. A research team from The Institute for Education Research at UHN, Carleton University, and York University explored public perspectives on organ transplantation using an arts-based approach to capture insights that may be missed by traditional research methods and support more patient-centred care.
To explore the social and emotional aspects of transplantation, researchers examined the use of art to understand people’s experiences and reflections on life after transplantation. To do this, the team hosted a series of public events, including art exhibitions, talks, conference sessions, and film screenings featuring stories, films, and artwork created by transplant recipients. Attendees were invited to reflect on the transplant-related events by creating their own drawings and designs using provided art supplies—a form of visual field notes.
Field notes are commonly used by researchers to document observations and better understand people’s experiences, practices, and cultures. In this study, the research team invited members of the public to contribute visual field notes, exploring a collective and accessible way of understanding the complexities of transplantation.
The research team analyzed the visual field notes, identifying four recurring themes. First, many of the pieces expressed difficult emotions, such as isolation, fear, and distress, often through the use of darker colours. Second, participants also used different materials and textures to represent physical and emotional experiences, such as loss or change. For example, a torn page was used to show organ removal or loss. Third, written and visual elements often represented internal conflicts about treatment and future outcomes. Lastly, many pieces challenged the idea that organ transplantation is a purely positive experience, highlighting how real-life experiences can be more complex.
The research team later assembled the pieces into a temporary, interactive sculpture and invited new participants to engage with it, encouraging discussion. This process revealed how perceptions of organ transplantation may change across different audiences.
Overall, the findings suggest that arts-based approaches may help deepen understanding of complex health experiences by capturing emotional and social aspects of organ transplantation that are often overlooked. By bringing these perspectives into research and care discussions, this work may help support more comprehensive, patient-centred care.
Dr. Kelly Fritsch, first author of the study, is an Associate Professor in the Department of Sociology and Anthropology at Carleton University. Dr. Fritsch is also a TIER 2 Canada Research Chair in Disability, Health, and Social Justice.
Dr. Suze Berkhout, senior author of the study, is an Education Investigator at The Institute for Education Research at UHN. Dr. Berkhout is also an Associate Professor in the Department of Psychiatry and Affiliate Faculty at the Institute for the History and Philosophy of Science and Technology at the University of Toronto.
This work was supported by UHN Foundation, the New Frontiers Research Fund, the Social Sciences and Humanities Research Council of Canada, and the Canadian Donation and Transplantation Research Program.
Fritsch K, Park M, Banerjee T, Berkhout S. Experimental Afters: On Meaning-Making with Collective Visual Fieldnotes. Leonardo. 2026 Apr 1. doi: 10.1162/LEON.a.2634.

Examples of the visual field notes created by participants. (Images courtesy of Dr. Suze Berkhout)
×
Surgical Research Excellence
UHN researcher awarded prestigious surgical research honour.

Dr. Mojgan Hodaie, Senior Scientist at the UHN’s Krembil Brain Institute, has been awarded the prestigious Lister Prize, the University of Toronto Department of Surgery’s highest research honour.
The award was presented at the 2026 Department of Surgery Gallie Day and Gordon Murray Banquet, an annual event that honours the outstanding achievements within the surgical community. The Lister Prize recognizes investigators who demonstrate sustained, internationally recognized excellence through research publications, grants, mentorship, and leadership.
This accolade reflects Dr. Hodaie’s dedication to advancing surgical research while maintaining an active clinical and academic practice. As a respected neurosurgeon and scientist, she also plays a key role in mentoring the next generation of clinicians and scientists. At UHN, Dr. Hodaie leads a robust research program focused on understanding and treating chronic neuropathic pain. Using advanced imaging techniques, her work is helping transform patient care and improve outcomes.
At the University of Toronto, Dr. Mojgan Hodaie is a Professor in the Department of Surgery. Read more about the Gallie Day awards here.
×
The Science Behind Safer Eating
Testing different food textures can support better care for people living with dysphagia.
Read MoreThe Science Behind Safer Eating
Testing different food textures can support better care for people living with dysphagia.

Image Caption: Prof. Catriona Steele, senior author of the study, pictured here analyzing a videofluoroscopy recording. This technique allows clinicians to observe swallowing in real time and gain insights to better guide dysphagia care.
People with swallowing difficulties, known as dysphagia, often rely on texture-modified foods, such as purées and thick liquids, to make eating safer and easier. However, these changes can reduce nutritional value and enjoyment, highlighting the need to reassess current dietary recommendations. Researchers from UHN’s KITE Research Institute found that different food textures can influence measures of swallowing timing and efficiency, offering new data to inform clinical assessments and care.
Using videofluoroscopy—a moving X-ray that shows how food travels through the throat in real time—the research team studied 20 healthy adults as they ate foods of different textures, as defined by the International Dysphagia Diet Standardisation Initiative: minced and moist, soft and bite-sized, and regular solid food. The team measured how quickly the food moved through the throat, how effectively the throat muscles worked, and how much food was left behind (residue). They also assessed safety by checking whether any food entered the airway.
The findings showed that swallowing performance changed with texture. Foods with larger particles that required more chewing moved through the throat more slowly. Minced and moist foods moved the fastest, followed by soft and bite-sized foods, and then regular solids. Residue left behind after swallowing was highest for minced and moist and regular solids, and lowest for soft and bite-sized foods. Regardless of food texture, all participants swallowed safely, with no food entering the airway.
Importantly, the findings showed that evaluations of swallowing with thick liquids and purées are insufficient to predict how a person will swallow food. These smoother textures do not reflect how people eat in everyday life. This is a key finding as current assessments do not always include food. The researchers recommend that a solid food item, like a cracker, be routinely included in swallowing assessments to fully understand a person’s swallowing efficiency and muscle function.
Overall, the study suggests that solid food should be included in swallowing assessments to inform optimal diet texture recommendations. Although further research is needed in clinical populations, these findings may help guide more balanced approaches to dysphagia management that support safety and quality of life.
Pooja Gandhi, first author of the study, was a former PhD student in the lab of Prof. Catriona Steele. She is currently an Assistant Professor in the Department of Communication Sciences and Disorders at the University of Alberta.
Prof. Catriona Steele, senior author of the study, is a Senior Scientist at UHN’s KITE Research Institute and a Tier 1 Canada Research Chair in Swallowing and Food Oral Processing. She is also a Professor in the Department of Speech-Language Pathology and a Faculty Member of the Rehabilitation Sciences Institute at the University of Toronto
This work was supported by UHN Foundation, the National Institute on Deafness and Other Communication Disorders, and the National Institute on Aging.
Prof. Steele is the developer of the ASPEKT Method of videofluoroscopy analysis, which was used in this study. She was also a board member of the International Dysphagia Diet Standardisation Initiative from 2012 to 2023.
Gandhi P, Barrett E, Mancopes R, Panes V, Peladeau-Pigeon M, Simmons MM, Steele CM. What About Foods? The Influence of Food Texture on the Safety, Timing, Kinematics, and Efficiency of Pharyngeal Phase Swallowing in Healthy Adults. J Speech Lang Hear Res. 2026 Apr 10. doi: 10.1044/2025_JSLHR-25-00546.
×

More Biomarkers Improve Detection
New diagnostic approach combining skin and blood tests could overhaul Parkinson disease care.
Read MoreMore Biomarkers Improve Detection
New diagnostic approach combining skin and blood tests could overhaul Parkinson disease care.

Image Caption: When interpreted together, three biomarkers—4-repeat tau (4R-tau), neurofilament light chain (NfL), and α-synuclein—can help distinguish between Parkinson disease (PD) and related conditions. α-synuclein is commonly found in PD and multiple system atrophy (MSA), while 4R-tau is strongly associated with progressive supranuclear palsy (PSP). Higher levels of NfL in the blood reflect greater nerve cell damage and can help distinguish more aggressive conditions like MSA from PD. Together, these markers provide a more accurate picture than any single test alone.
With the emergence of new disease-modifying therapies that target the specific proteins responsible for neurodegenerative diseases like Alzheimer disease and Parkinson disease (PD), the timing and accuracy of diagnosis are increasingly important. Published in Nature Medicine, a new study led by Drs. Anthony Lang and Ivan Martinez-Valbuena at UHN’s Krembil Brain Institute (KBI), introduces a diagnostic approach that combines multiple biomarkers—biological molecules, often proteins, that can signal disease—to better distinguish between PD and similar neurodegenerative conditions called parkinsonian syndromes. This approach could support earlier and more accurate diagnosis, and better care for patients as a result.
The challenge in getting it right
Current diagnostic methods often misclassify other parkinsonian syndromes, including progressive supranuclear palsy (PSP) and multiple system atrophy (MSA), as PD because of overlapping symptoms. This challenge is especially pronounced in early disease stages, when distinguishing clinical features are not yet present.
However, despite their similarities, these conditions are driven by different underlying proteins—and identifying which is key to improving diagnosis.
“Misdiagnosis means patients miss the opportunity to use disease-modifying therapies during the critical window in early disease stages when these treatments may have the greatest impact,” says Dr. Martinez-Valbuena, the first author of this study.
A new approach: combining multiple biomarkers
To address this issue, the KBI team developed a diagnostic protocol that integrates three biomarkers associated with parkinsonian syndromes: 4-repeat tau (4R-tau), neurofilament light chain (NfL), and α-synuclein. Each biomarker provides distinct information. α-synuclein identifies the abnormal protein build up seen in diseases like PD and MSA. 4R-tau signals the abnormal protein build up seen in PSP. NfL reflects the extent of damage to nerve cells. The new protocol uses a blood sample to measure NfL, and a single skin biopsy is to perform tests called seeding amplification assays (SAAs) to identify 4R-tau and α-synuclein. Although evaluation of α-synuclein is currently marketed diagnosis of PD, these diagnostic methods do not use SAAs.
The researchers first tested their approach in a sample of 166 participants—40 with PD, 77 with PSP, 29 with MSA, and 20 healthy controls. They then validated their findings in an independent cohort of 63 participants—35 with PD, 17 with PSP, nine with MSA, and two healthy controls. Participants in both groups varied in age and duration since disease onset.
Individually, each biomarker test showed high sensitivity and specificity. Skin tests detected α-synuclein in approximately 85% and 90% of people with PD and MSA, respectively, and 4R-tau in about 88% of people with PSP. Blood levels of NfL were lowest in healthy individuals and highest in people with MSA. The multimodal approach significantly outperformed the diagnostic accuracy—herein defined as the ability to correctly distinguish between MSA, PSP, and PD—of any single biomarker test on its own; receiving a performance score (AUC) of 0.96 for PD, 0.90 for MSA and 0.97 for PSP. AUC describes how well a model, the diagnostic protocol in this case, matches the data it is trying to explain. A perfect model has an AUC of 1.
Importantly, α-synuclein was found in about 23% of patients with PSP. This indicated that diagnostic testing that is limited to this protein, as some companies currently offer, is insufficient as it could misdiagnose PSP as PD.
Notably, the researchers also found that, in addition to better differentiating between PSP and other parkinsonian syndromes, combining biomarkers also enabled doctors to separate patients according to PSP severity.
To further validate the method, the KBI team compared the results from their diagnostic protocol with findings from brain tissue analysis in a subset of 11 participants. The diagnosis made using the integrated protocol consistently matched brain tissue findings, providing strong real-world validation of the approach. Importantly, in 1 case clinically diagnosed as having PSP—where the individual’s skin SAA tested negative both 4R tau and α-synuclein—brain tissue analysis later revealed a diagnosis of Alzheimer disease. This alternative diagnosis validated the negative skin SAA results.
A promising (and less invasive) path forward
While researchers emphasize that further testing is required—particularly in patients in earlier disease stages—the findings represent a promising advancement in the diagnosis of parkinsonian syndromes.
“Combining assessment of 4R-tau, NfL, and α-synuclein was a logical step,” says Dr. Lang, the study’s senior author. “These biomarkers complement each other—where one shows limitations, the others provide additional diagnostic value.”
“We are encouraged by the potential of this protocol to address gaps in the clinical evaluation of complex or unclear parkinsonian syndromes,” adds Dr. Martinez-Valbuena.
In addition to performance, the KBI team’s new diagnostic approach is also advantageous because it is less invasive than other approaches. The integrated approach uses a blood sample and one skin biopsy. This contrasts currently marketed testing for α-synuclein, which requires a cerebrospinal fluid sample obtained through a lumbar puncture or multiple skin biopsies. By simplifying sample collection and reducing reliance on specialized equipment and procedures, the integrated diagnostic protocol may make diagnostic testing more widely accessible.
For patients with PD, MSA, and PSP, earlier and more precise diagnosis could enable better disease management and, eventually, an improved quality of life. As new therapies continue to emerge, tools like this may help ensure patients receive the right care at the right time.
The first author of this study, Dr. Ivan Martinez-Valbuena, is a Scientific Associate at UHN’s Krembil Brain Institute.
The senior author of this study, Dr. Anthony E. Lang, is a Senior Scientist at UHN’s Krembil Brain Institute and a Professor at the University of Toronto's Temerty Faculty of Medicine and Institute of Medical Science. He is also the Lily Safra Chair in Movement Disorders, the Director of the Edmond J. Safra Program in Parkinson's Disease, Director of the Rossy PSP Centre, and the Jack Clark Chair for Parkinson’s Disease Research.
This work was supported by the Rossy Family Foundation, the Edmond J. Safra Philanthropic Foundation, the Maybank Foundation, the Blidner Family Foundation, Ajay Virmani, Jen Cerny, the Michael J. Fox Foundation for Parkinson’s Research., the Rainwater Charitable Foundation, the Bay Tree Foundation/Mohammad Al Zaibak Fellowships in Parkinson’s Disease, The Paul and Susan Hansen Foundation, the University Medical Center Goettingen-UMG Clinician Scientist Program, the Mohammad and Najla Al Zaibak Family Parkinson’s Disease Research Fund, the Fonds de Recherche du Québec Santé, and UHN Foundation.
Drs. Martinez-Valbuena, Kovacs, and Lang share a pending patent for movement disorders diagnostic assays, and Dr. Kovacs has a patent for the 5G4 synuclein antibody.
Dr. Martinez-Valbuena received consulting fees from Ferrer and research funding from the Michael J. Fox Foundation for Parkinson’s Research outside of this work.
Dr. Lang served as an advisor for AbbVie, Amylyx, Aprinoia, Biogen, BioAdvance, Biohaven, BioVie, BlueRock Therapeutics, Bristol Myers Squibb, Denali, EG427, Ferrer, Janssen, Lilly, Northera, Pharma 2B, Sun Pharma, UCB and Ventyx Bio. He also received honoraria from Sun Pharma and AbbVie and grants from Brain Canada, the Canadian Institutes of Health Research, the Michael J. Fox Foundation for Parkinson’s Research, the Parkinson Foundation, Parkinson Canada, and the Weston Foundation. Dr. Lang also serves as an expert witness in litigation related to paraquat and PD, and has received publishing royalties from Elsevier, Saunders, Wiley-Blackwell, Johns Hopkins Press, and Cambridge University Press.
For a full list of competing interests, see the publication.
Martinez-Valbuena I, Emamikhah M, Olszewska DA, Weber SK, Schnell S, Fereshtehnejad SM, Reyes NGD, Sousa M, Di Luca DG, Ta J, Anastassiadis C, Li J, Sasitharan J, Bhakta P, Visanji NP, Fox SH, Mollenhauer B, Tartaglia MC, Kovacs GG, Lang AE. Dermal α-Synuclein and 4R-Tau SAAs Combined with Serum NfL: Enhancing Diagnostic Precision in Neurodegenerative Parkinsonism. Nat Med. 2026 May 19. doi: 10.1038/s41591-026-04398-3
×

Sustaining Global Cancer Education
Study finds that sustainable cancer education requires connection and institutional support.
Read MoreSustaining Global Cancer Education
Study finds that sustainable cancer education requires connection and institutional support.

Image Caption: Global oncology is a field of research focused on improving cancer care worldwide by addressing disparities in resources and expertise across diverse health systems.
The Global Oncology Enrichment Program (GOEP) is an online education program designed to address inequities in access to cancer education for clinicians worldwide. Researchers from The Institute for Education Research (TIER) at UHN evaluated the program’s impact, examining how different delivery formats can influence learning about global oncology topics and levels of engagement among clinicians.
GOEP is hosted on UHN’s Princess Margaret Cancer Campus, which serves as a hub for cancer education for clinicians and trainees. Launched in 2020, the program combined live and recorded webinars with interactive Q&A sessions. Course topics included global oncology challenges and opportunities, cancer prevention and screening, advocacy, research, and policy. In 2022, the program transitioned to a fully self-directed format, offering recorded webinars to improve accessibility and reduce the administrative demands associated with delivering live sessions.
To better understand GOEP’s impact across the two delivery formats, TIER researchers analyzed responses from 256 program participants who completed pre- and post-course surveys assessing motivations and barriers to accessing global oncology education, as well as confidence in key topic areas. The analysis showed that participants’ confidence in their knowledge of global oncology topics increased in both the combined live and recorded format and the self-directed format. However, levels of engagement differed between the two formats.
Participants in the combined format were more likely to complete the program compared to those in the self-directed format and identified opportunities for networking, mentorship, and career exploration as key motivators for participation. However, participants in the combined format also reported limited faculty interest in and support for global oncology at their respective institutions. These findings highlight a gap between having access to global oncology education and the institutional support available for career development.
After the program transitioned to the self-directed format, overall participation and completion rates declined. Participants who remained engaged were more likely to be radiation oncologists, radiation therapists, or individuals already involved in global oncology initiatives.
The results of this study underscore the importance of providing accessible education alongside institutional support and opportunities for connection to promote equity, sustained engagement, and real-world impact in global cancer care.
Dr. Revadhi Chelvarajah, co-first author of the study, was a previous clinical fellow in the Department of Radiation Oncology at UHN’s Princess Margaret Cancer Centre.
Tina Papadakos, co-first author of the study, is an Education Investigator 1 at The Institute for Education Research at UHN and the Director of Knowledge Translation with Cancer Education at UHN’s Princess Margaret Cancer Centre.
Dr. Meredith Giuliani, senior author of the study, is an Education Investigator 2 at The Institute for Education Research at UHN. At the University of Toronto, Dr. Giuliani is the Associate Dean of Postgraduate Medical Education.
This work was supported by The Princess Margaret Cancer Foundation.
Dr. Giuliani has a leadership role at the Internationals Journal of Radiation Oncology Biology Physics. See the article for the full list of author disclosures.
Chelvarajah R, Papadakos T, Anakwenze C, Bourque JM, Chaudhary S, Choi S, Kizub D, Liu ZA, Papadakos J, Storer S, Walker E, Ye XY, Rodin D, Giuliani M. Creating Sustainable Education Programming to Build Capacity for Collaborative Global Cancer Control. JCO Glob Oncol. 2026 Mar 19. doi: 10.1200/GO-25-00372.
×
Research Institutes at UHN
Research conducted at UHN's research institutes spans the full spectrum of diseases and disciplines, including cancer, cardiovascular sciences, transplantation, neural and sensory sciences, musculoskeletal health, rehabilitation sciences, and community and population health.
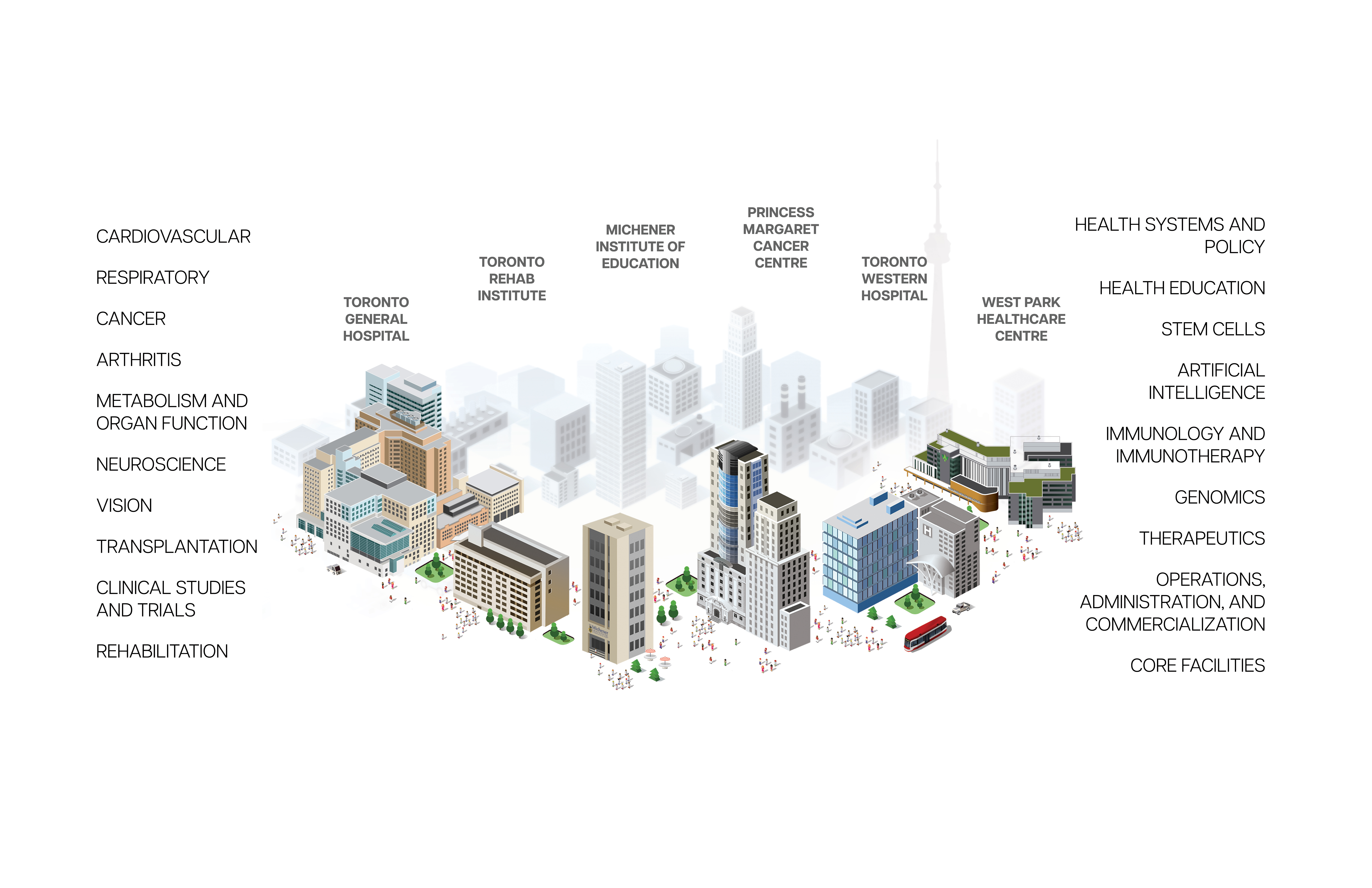
Research Institutes
Research at UHN is conducted under the umbrella of the following research institutes. Click below to learn more:





