Achieving Institutional Authorization
To begin the IA process, all research submissions must be entered into the Coordinated Approval Process for Clinical Research (CAPCR)*, which is a web-based application system through which researchers can obtain all the approvals required for conducting research involving humans at UHN. CAPCR can be accessed while connected to the research network or via the Remote Login page.
Summary of Administrative Process
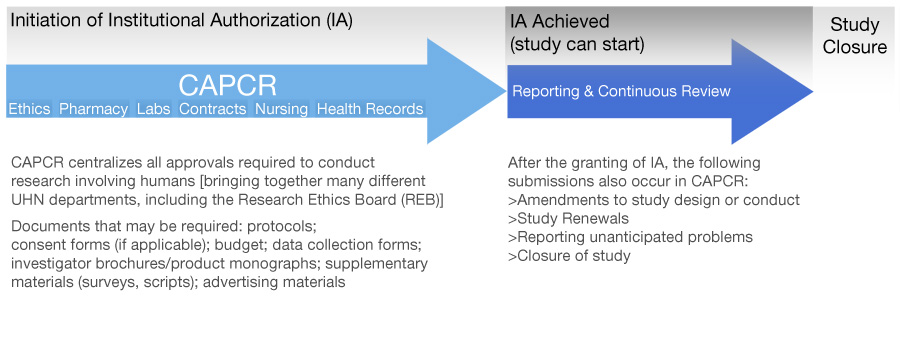
For more information about the IA process, contact Clinical Research Support Systems (CRSS).
For more information about the ethics review process, see the Ethics Contact page.
Understanding Institutional Authorization
All research involving humans performed at UHN requires Institutional Authorization (IA) prior to the conduct of the research. IA is granted on a per-protocol basis by the Executive Vice President of Science and Research or designate once all requisite approvals have been obtained.
The determination of whether a particular activity requires IA depends on two factors:
If both the conditions above are met (ie, you wish to conduct research involving humans and UHN would be engaged in the research), IA is required.