
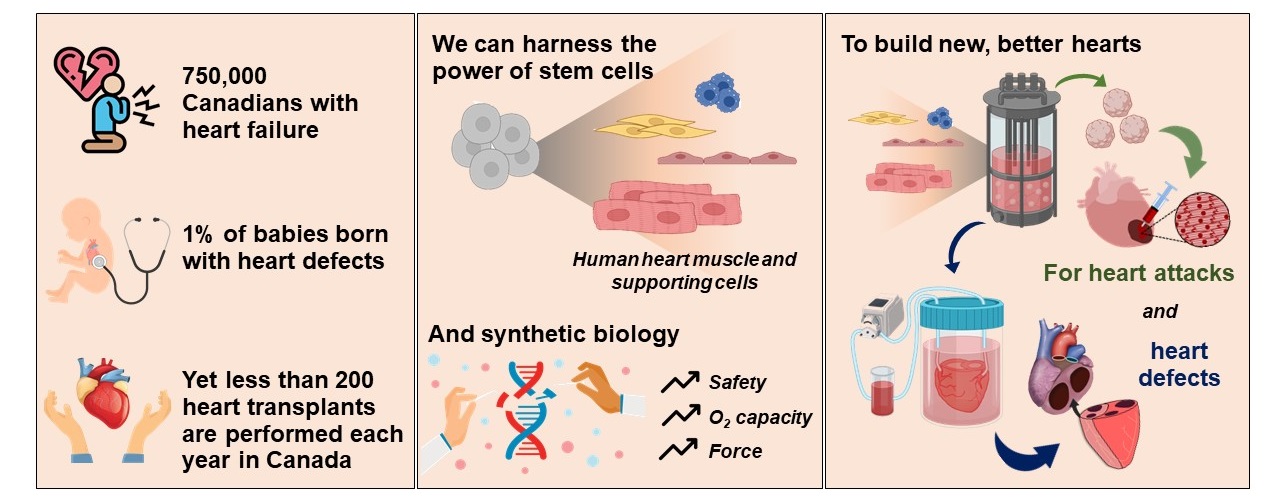
Ischemic heart disease remains the leading cause of death in Canada and worldwide. Modern medical management has improved the prognosis of patients after myocardial infarction (MI), but existing therapies are largely aimed at slowing disease progression rather than restoring lost contractile function. My laboratory is focused on developing novel therapies for post-MI heart failure based on human pluripotent stem cells (hPSCs) because hPSCs are the only stem cell type capable of differentiating into large quantities of phenotypically unambiguous cardiomyocytes. Our goal is to restore the electrical and contractile function of injured hearts by “remuscularizing” the infarct scar with hPSC-derived cardiomyocytes.
Our laboratory has already made a number of important advances in this area, including the development of efficient protocols to guide hPSCs into cardiomyocytes and specialized cardiac subtypes, proof-of-concept transplantation studies with hPSC-derived cardiomyocytes in rodent MI models, and the first direct demonstration that hPSC-derived cardiomyocytes can become electrically integrated and activate synchronously with host myocardium in injured hearts. Our ongoing work builds on these successes and is bringing us closer to a viable cell therapy. Current projects in the lab include 1) developing scalable approaches to promote the maturation of hPSC-derived cardiomyocytes in vitro, 2) creating and validating new tools to characterize the electrical behavior of hPSC-derived cardiac grafts in vivo, 3) exploring novel pharmacological and gene-engineering approaches to improve host-graft electromechanical integration and graft electrophysiological function, and 4) testing the efficacy and safety of hPSC-derived cardiomyocyte transplantation in highly relevant preclinical MI models.
Our laboratory has already made a number of important advances in this area, including the development of efficient protocols to guide hPSCs into cardiomyocytes and specialized cardiac subtypes, proof-of-concept transplantation studies with hPSC-derived cardiomyocytes in rodent MI models, and the first direct demonstration that hPSC-derived cardiomyocytes can become electrically integrated and activate synchronously with host myocardium in injured hearts. Our ongoing work builds on these successes and is bringing us closer to a viable cell therapy. Current projects in the lab include 1) developing scalable approaches to promote the maturation of hPSC-derived cardiomyocytes in vitro, 2) creating and validating new tools to characterize the electrical behavior of hPSC-derived cardiac grafts in vivo, 3) exploring novel pharmacological and gene-engineering approaches to improve host-graft electromechanical integration and graft electrophysiological function, and 4) testing the efficacy and safety of hPSC-derived cardiomyocyte transplantation in highly relevant preclinical MI models.
Enabling novel heart therapies with stem cells
Study Status: active
Institute: McEwen Stem Cell Institute
Health Conditions:
Heart disease and disorders
- Robert McEwen Chair in Cardiac Regenerative Medicine, University Health Network
- Staff Pathologist, University Health Network
- Associate Professor, Laboratory Medicine and Pathobiology, University of Toronto