University Health Network (UHN) is a research hospital affiliated with the University of Toronto and a member of the Toronto Academic Health Science Network. The scope of research and complexity of cases at UHN have made it a national and international source for discovery, education and patient care.
Recent News

AI for Transplant Decision-Making
Researchers highlight the potential of AI agents to support health care decision-making.
Read MoreAI for Transplant Decision-Making
Researchers highlight the potential of AI agents to support health care decision-making.

Image Caption: Large language models (LLMs), a type of artificial intelligence, have shown success in health care-related tasks such as assessing surgical risks and summarizing health records. LLMs can also be used as ‘agents’ that perform specialized tasks. This approach has the potential to help tackle complex health care decisions—such as liver transplant selection.
Researchers from UHN’s Ajmera Transplant Centre have simulated an organ transplant selection committee using an AI framework. The study, published in The Lancet Digital Health, found that an AI system made up of several specialized components, called “agents”, could accurately identify which patients were likely to benefit from a liver transplant and which were not.
Liver transplantation is offered as a life-saving intervention for patients with end-stage liver disease. However, the demand for organs often exceeds the supply available for transplantation. Candidacy for a transplant is considered based on need and survival benefit for patients.
Decisions about which patients should be placed on liver transplant waiting lists are currently made by multidisciplinary transplant selection committees. Despite established guidelines to ensure fairness, equitable access, and standardization, as well as the best intentions of clinicians involved, subjectivity and bias can still impact the process. This can result in variability in decisions within and between institutions, especially for complex cases. Therefore, there is a need for greater objectivity and consistent, data-driven decision-making.
Large language models (LLMs)—AI systems trained on large amounts of text that can read, interpret, and generate human-like language—are increasingly being used as tools in health care. LLMs can act as AI agents—systems capable of autonomously performing tasks on behalf of a user. They can also be used in multi-agent collaborations, where multiple AI agents work collectively to help solve complex problems.
Although multi-agent frameworks have the potential to support health care decision-making for clinical teams, their application in the medical field remains limited.
To investigate the use of multi-agent AI as a way to aid in objective health care decision-making, the researchers examined the performance of a multi-agent AI selection committee (AI-SC) in the liver transplant selection process. This AI committee was designed to reflect a typical transplant team, with each AI agent assigned the role of one member of the team, including a transplant hepatologist, transplant surgeon, cardiologist, or social worker.
The AI-SC then assessed whether patients should be added to the transplant waiting list by determining whether a liver transplant was likely to improve survival at six months or one year. Patients were declined if medical factors made transplantation inappropriate to consider or unlikely to extend life.
The AI-SC was tested using information from a database of approximately 8,400 liver transplant candidates from 2004 to 2023. Researchers analyzed the outcomes of patients who received a transplant, and then created a simulated group of patients, including patients with medical conditions that would automatically rule out transplantation, for the AI-SC to assess.
Results showed that the AI-SC triaged potential transplant patients with high accuracy. It correctly identified patients who should not receive a transplant 98% of the time, and predicted whether a transplant would provide a survival benefit at both six months and one year after surgery with accuracies of 92% and 95%, respectively.
There were still instances where the AI-SC declined patients who had ultimately been accepted (false negatives) or accepted patients who died before 6 months or a year post-transplant (false positives). These false negatives and false positives were largely related to underlying cancer‑related factors and greater complexity in the patients' medical history.
Overall, the findings suggest that multi-agentic AI could potentially support transplant teams by helping them make consistent, evidence‑based decisions—improving the transplant selection process. This UHN-developed tool is the first instance of a multi-agentic AI system to guide objective, consensus-based decision-making in health care. As an important next step, the Transplant AI Initiative team is currently evaluating this tool at an international, multicenter level to assess its role across different clinical contexts.
Dr. Bima Hasjim is an International Clinical Fellow in the Bhat lab and a surgical resident at the University of California. He is a co-first author of the study.
Dr. Ghazal Azarfar is a Research Associate in the Bhat lab and co-first author of the study.
Dr. Divya Sharma is an AI Scientist and Clinician Investigator at UHN’s Princess Margaret Cancer Centre. She is an Assistant Professor at York University in the Department of Mathematics and Statistics, with an additional appointment at the Dalla Lana School of Public Health at the University of Toronto. She is a co-senior author of the study.
Dr. Mamatha Bhat, Scientist at UHN’s Ajmera Transplant Center and Associate Professor in the Department of Medicine at the University of Toronto, is a co-senior author of this study.
This work was supported by the Transplant AI Initiative, Ajmera Transplant Centre, and UHN Foundation.
Dr. Bhat has received grants/contracts from Novo Nordisk, Paladin, Oncoustics, Merck, and Transplant Genomics, payment/honoraria from Paladin, and reports participating on a Data Safety Monitoring Board/Advisory Board for Novo Nordisk.
Hasjim BJ, Azarfar G, Lee FG, Diwan TS, Raju S, Gross JA, Sidhu A, Ichii H, Krishnan RG, Mamdani M, Sharma D, Bhat M. A multiagent large language model–based system to simulate the liver transplant selection committee: a retrospective cohort study. Lancet Digit Health. 2026 Apr 7;100966. doi:10.1016/j.landig.2025.100966. Epub 2026 Apr 7.
×

Investing in Research Impact
UHN receives $8.6 million in funding from the Canada Foundation for Innovation.
Read MoreInvesting in Research Impact
UHN receives $8.6 million in funding from the Canada Foundation for Innovation.

Image Caption: (Pictured clockwise from the top left) Drs. Michael Laflamme, Benjamin Haibe-Kains, and Hui Peng. (Images: UHN and SGC)
The Government of Canada has announced an investment of over $552 million to support high-impact research infrastructure projects through the Canada Foundation for Innovation (CFI). Delivered through CFI’s Innovation Fund, this major investment supports 92 research infrastructure projects at 32 institutions across the country—including four at UHN.
UHN researchers were awarded over $8.6 million in funding across two UHN-led projects and two projects led by collaborating institutions. The following UHN-led research teams received funding for large-scale infrastructure-supporting projects on advanced preclinical models and AI-driven drug discovery:
Dr. Michael Laflamme | Senior Scientist, UHN’s McEwen Stem Cell Institute
Dr. Laflamme is the lead on a project that received $3.4 million for enabling advanced preclinical modelling for the translation of biomedical innovations. By enhancing these models, researchers aim to strengthen the evaluation of new therapies, medical devices, and complex procedures, helping move promising discoveries safely toward patient care. UHN project members include Drs. Alyssa Goldstein, Christoph Haller, Shaf Keshavjee, Sonya MacParland, Kumaraswamy Nanthakumar, M. Cristina Nostro, Stephanie Protze, Paul Santerre, and Sara Vasconcelos.
The University of Toronto (U of T) is the collaborating institution on this project. At U of T, Dr. Laflamme is a Professor in the Department of Laboratory Medicine and Pathobiology.
Dr. Benjamin Haibe-Kains| Senior Scientist, UHN’s Princess Margaret Cancer Centre & Dr. Hui Peng | Affiliate Scientist, UHN’s Princess Margaret Cancer Centre
Drs. Haibe-Kains and Peng are co-leading a project that received $2.7 million to advance drug discovery through the generation of chemical data and the development of AI models shared on a new cloud-based open platform, AIRCHECK. This project combines advanced experimental technologies with artificial intelligence to make drug discovery faster, more efficient, and more accessible to researchers worldwide. UHN project members include Drs. Cheryl Arrowsmith, Levon Halabelian, Rachel Harding, Matthieu Schapira, and Bo Wang. Dr. Haibe-Kains is also an Associate Professor in the Department of Medical Biophysics and an Adjunct Professor in the Department of Computer Science at U of T. Dr. Peng is an Associate Professor in U of T's Department of Chemistry.
UHN also received $2.4 million through projects led by collaborating institutions. Dr. Arash Zarrine-Afsar, Senior Scientist at UHN’s Princess Margaret Cancer Centre (PM), is the co-lead on a project developing a comprehensive image-guided biomarker discovery platform. Dr. Michael Brudno, Senior Scientist at PM, is a collaborating researcher on a project focused on gathering vital real-time health data for clinical trials, artificial intelligence, and a learning health system, also known as the VITAL project.
Congratulations to all UHN award recipients. Read a full list of funded projects here.
×

A Bridge Beyond Pacemakers
UHN team generates cardiac-like cells that could replace electronic pacemakers after AV block.
Read MoreA Bridge Beyond Pacemakers
UHN team generates cardiac-like cells that could replace electronic pacemakers after AV block.

Image Caption: The cover image of Cell Stem Cell depicts the heart’s conduction system as a highway, with cars symbolizing electrical signals. The cars are crossing a damaged AVN bridge representing AV block. A new innovation using heart cells grown in the lab acts as a bandage to restore this passage. Illustration by Maggie Kwan; Cover Cell Stem Cell ©2026.
A research team from UHN’s McEwen Stem Cell Institute (McEwen) has grown heart cells in the lab that mimic the atrioventricular node (AVN), the part of the heart that carries electrical impulses from the atria to the ventricles. This innovation could offer individuals with a dysfunctional AVN a novel treatment option—called a biological conduction bridge—that may one day replace standard pacemaker implants.
As the “electrical bridge” between the atria and ventricles, the AVN is responsible for maintaining the pace of the ventricles’ contractions and, thus, efficient blood flow. An improperly functioning AVN can lead to a life-threatening condition called atrioventricular (AV) block, which is typically treated by implanting an electronic pacemaker (EPM) to take over the AVN’s function. Although lifesaving, EPMs are not without limitations, including the need for surgery to replace their batteries every 10 to 15 years and their inability to respond to physiological changes. These limitations underscore the need for alternative approaches that more closely mimic native cardiac conduction.
In pursuit of an alternative, cell-based therapy for AV block, Dr. Stephanie Protze, a McEwen Scientist, and her team used a specialized lab process that turns stem cells into specific cell types to create cardiac pacemaker cells that behave like those in the AVN (AVNLPCs). The team then assessed gene expression and functional properties to confirm the cells’ identity as AVNLPCs.
The AVNLPCs showed the same key biological markers as AVN pacemaker cells in developing human hearts. For example, the cells showed high expression of key AVN genes, including TBX3, MSX2, RSPO3, and low expression of genes such as SCN5A, GJA1, GJA5, which are characteristic of other cell types, such the heart muscle cells.
When integrated into a 3D–heart tissue model, the AVN-like pacemaker cells’ responses to electrical signals were comparable to those observed in the human heart. This includes slow electrical impulse conduction and blocking conduction of fast atrial rates, such as those seen in atrial fibrillation, which would be life-threatening in the ventricles.
Importantly, when the researchers implanted their AVNLPCs into a preclinical model of SAN and AVN dysfunction, the cells remained functional and continued to display the same electrophysiological properties observed in the 3D–heart tissue model.
Overall, this work establishes a foundation that could be used to further develop a biological conduction bridge. The McEwen team’s work offers the first evidence that AVNLPCs are functional in multiple model types, both in vitro and in vivo. Importantly, they demonstrate that AVNLPCs mimic the key safety features of AVN conduction in humans—namely, the ability to block fast atrial rates from reaching the ventricles, which is a key safety feature of the AVN.
Although additional studies are required to replicate these results and test the efficacy of AVNLPCs in additional lab models, and eventually in humans, this work represents an important step toward a more biologically relevant and durable alternative to electronic pacemakers for patients with AV block.
The first author of this study is Dr. Michelle Lohbihler, a former graduate student at UHN’s McEwen Stem Cell Institute in the Protze Lab.
The senior author of this study is Dr. Stephanie Protze, a Scientist at UHN’s McEwen Stem Cell Institute and an Assistant Professor of Molecular Genetics at the University of Toronto’s Temerty Faculty of Medicine.
Drs. Sara Nunes Vasconcelos, Michael Laflamme, and Kumaraswamy Nanthakumar, who are Senior Scientists at UHN, also contributed to this study. Dr. Laflamme is also a Tier 1 Canada Research Chair in Cardiovascular Regenerative Medicine.
This work was supported by the Canadian Institutes of Health Research, the Canadian Foundation for Innovation, the Government of Canada’s New Frontiers in Research Fund, the Canada Research Chairs Program, BlueRock Therapeutics, and UHN Foundation.
Dr. Laflamme receives funds as a consultant for and is also a scientific founder of BlueRock Therapeutics. Drs. Laflamme and Protze also share a patent for materials related to this study.
Lohbihler M, Lim AA, Massé S, Kwan M, Mourad O, Mastikhina O, Murareanu BM, Elbatarny M, Sarao R, Qiang B, Dhahri W, Chang ML, Xu ALY, Mazine A, Khattak S, Nunes SS, Nanthakumar K, Laflamme MA, Protze S. Human pluripotent stem cell-derived atrioventricular node-like pacemaker cells exhibit biological conduction bridge properties in vitro and in vivo. Cell Stem Cell. 2026 Mar 23. doi: 10.1016/j.stem.2026.02.012.
×

New Models of Cardiac Rehabilitation
Hybrid and virtual care models are expanding access and improving patient-centred cardiac care.
Read MoreNew Models of Cardiac Rehabilitation
Hybrid and virtual care models are expanding access and improving patient-centred cardiac care.

Image Caption: The COVID-19 pandemic transformed how cardiac care was delivered worldwide, highlighting the need to understand its impact on program delivery and care models.
Before the COVID-19 pandemic, cardiac rehabilitation programs were traditionally delivered in person. However, when the pandemic began, programs rapidly shifted to virtual formats to maintain patient care. In a new study from UHN’s KITE Research Institute (KITE), researchers examined how this transition reshaped cardiac rehabilitation across Canada, expanding access and supporting more patient-centred models of care.
The research team surveyed 108 representatives from 150 cardiac rehabilitation programs to understand the barriers and benefits of in-person, virtual, and hybrid program models. Compared to programming before the pandemic, they found a significant decline in in-person programming and an increase in virtual and hybrid models. Close to 50% of rehabilitation programs now offer two or three delivery models.
The integration of hybrid and virtual models helped programs reach more participants. Virtual programs require fewer operational resources than in-person programs, which allow programs to support more patients without exceeding capacity. At the same time, virtual programs reduce financial or logistic barriers for patients who face challenges attending in-person, such as transportation costs, limited mobility, or restrictive schedules.
By directing lower-risk patients to virtual or hybrid formats, programs can allocate limited in-person resources to high-risk patients who require more supervised, onsite care, addressing long-standing eligibility restrictions. Ultimately, program selection was based on patient preference and collaborative discussions between patients and clinicians, enabling more tailored, patient-centred decision making.
However, in-person models remain important for social connectedness and peer support. Participants noted that patients are generally less satisfied with fully virtual formats. Researchers also found that patients with language or communication barriers face challenges across all models, particularly in person, where translation services were not consistently available.
With continued improvements in technology and better translation services, these evolving models could strengthen Canada’s cardiac rehabilitation system and enable more equitable, patient-centred care.
Dr. Susan Marzolini, lead author of the study, is a Scientist at UHN’s KITE Research Institute. At the University of Toronto, Dr. Marzolini is an Associate Professor at the Rehabilitation Sciences Institute and an Assistant Professor in the Faculty of Kinesiology & Physical Education.
This work was supported by UHN Foundation.
Marzolini S, Ocampo G, Hébert AA, Barbieri R, Cotie L, Barry-Hickey D, Martinuzzi M, Konidis R, Oh P. The Evolution of Cardiac Rehabilitation Since COVID-19. Heart Lung Circ. 2026 Feb 5. doi: 10.1016/j.hlc.2025.09.006.
×

Rehumanizing Classroom Care
Human-centred clinical letters can help schools better support children with disabilities.
Read MoreRehumanizing Classroom Care
Human-centred clinical letters can help schools better support children with disabilities.

Image Caption: Many children with disabilities require individualized school supports, yet disjointed communication between clinicians and educators can make that support difficult to obtain.
Children with disabilities often struggle to access appropriate school supports. Clinical letters are commonly used by clinicians to communicate medical information, management plans, and care recommendations to schools. A new study from The Institute for Education Research (TIER) at UHN highlights the importance of using a critically reflective approach when writing clinical letters to improve communication and collaboration between clinicians, families, and schools.
Clinical letters are a key form of communication between clinicians and schools. However, clinicians may be unaware of the everyday challenges that shape school-based care, such as limited resources and rigid policies, which can hinder how effectively schools apply clinicians' recommendations.
A critically reflective approach encourages clinicians to reflect on their assumptions and consider the systemic barriers that families and schools face. It also supports writing in a more collaborative, human-centred way, guiding clinicians to describe each child by highlighting strengths and goals, not just challenges, and to use team-oriented language, such as “We would like to work with you to…”.
To investigate the impact of critically reflective writing, the research team interviewed 17 parents and educators across Ontario and assessed their responses to clinical letters written by clinicians after completing training in this approach.
Participants felt that the critically reflective letters rehumanized children with disabilities and promoted stronger communication and collaboration. Shifting from challenge-focused descriptions to highlighting the children’s strengths and abilities changed how educators perceived them. Participants felt this made it easier to understand children’s needs and could support more trusting, open communication between families and educators. Improved communication and collaboration could also create new opportunities for shared advocacy by helping families and educators build a common understanding of challenges and identify where change is needed.
Overall, more thoughtful, human-centred clinical writing can help bridge communication gaps between health and education systems, ultimately improving support for children with disabilities.
Dr. Victoria Boyd, first and corresponding author of the study, is a Scientific Associate at The Institute for Education Research (TIER) and the Centre for Advancing Collaborative Healthcare & Education (CACHE) at UHN.
Dr. Nicole Woods, co-author of the study, is a Senior Scientist and Director of TIER. Dr. Woods is also a Scientist at The Wilson Centre at UHN as well as an Associate Professor in the Department of Family and Community Medicine, the Department of Surgery, and the Institute of Health Policy, Management and Evaluation at the University of Toronto (U of T).
Dr. Stella Ng, senior author of the study, is a TIER Scientist, Scientist at The Wilson Centre, and Director of CACHE at UHN. At U of T, Dr. Ng is an Associate Professor in the Department of Speech-Language Pathology and a Faculty Member of the Rehabilitation Sciences Institutes.
This work was supported by UHN Foundation, the Spencer Foundation, the Social Sciences and Humanities Research Council, Dr. Boyd’s Kimel-Schatzky Fellowship, and Dr. Ng’s Ontario Ministry Early Researcher Award and Arrell Family Chair in Health Professions Teaching.
Boyd, VA, Woods, NN., Campbell, WN, Kumagai, AK, & Ng, SL. (Re)humanizing clinical documentation for disabled children: a cascade of potential outcomes of critically reflective practice. Disability & Society. 2026 Feb 01. DOI: 10.1080/09687599.2025.2536587
×

Smartwatches for Heart Health
New study highlights how smartwatches can detect early signs of worsening heart failure.
Read MoreSmartwatches for Heart Health
New study highlights how smartwatches can detect early signs of worsening heart failure.

Image Caption: Wearable technology, such as a smartwatch, is effective at gathering physiological data (like heart rate) from people passively, in their daily lives. This makes these technologies good candidates for monitoring symptoms of heart failure that are difficult to measure in traditional clinical settings. Illustration by Katie Yost ©2025
A new study from UHN, featured on the cover of Nature Medicine, shows that wearable technologies such as Apple Watch can help monitor heart failure by providing early signs of deteriorating health where medical attention may be needed.
Heart failure—when the heart does not pump blood adequately to meet the body’s needs—is a global health crisis that leads to hospitalizations, increased use of health care services, and reduced life expectancy. Despite recent medical advances, patients with heart failure still face a high risk of poor outcomes. This highlights the need to improve risk assessment and better guide timely interventions.
“For patients with heart failure, periods of stability are often interspersed with flare-ups of symptoms such as shortness of breath or fatigue. These episodes may require medical attention to prevent hospitalization and improve quality of life,” says Dr. Heather Ross(link is external), Clinician Investigator at UHN and co-senior author of the study. “However, risk assessments for heart failure patients often rely on scheduled clinical visits or evaluation tools that take measurements at only one point in time. They don’t account for the changing, episodic nature of heart failure.”
Therefore, doctors and clinicians need to find better ways of monitoring heart failure symptoms and predicting when medical intervention may be needed. Wearable technology, such as Apple Watch, can continuously track important health and fitness measures, such as heart rate and blood oxygen levels, making it an excellent candidate for monitoring. However, until recently, it has been unclear how these measurements might be used to understand day-to-day changes in people living with heart failure.
To investigate this, the research team, led by researchers at UHN’s Peter Munk Cardiac Centre and the Ted Rogers Centre for Heart Research, initiated the Ted Rogers Understanding Exacerbations of Heart Failure study (TRUE-HF) in collaboration with Apple in the fall of 2020. The study examined whether biometric data from Apple Watch could be used to predict peak oxygen uptake (pVO2) in patients with heart failure in their daily lives for a three-month period. PVO2 is the highest amount of oxygen the body uses during intense exercise, serving as a key indicator of cardiorespiratory fitness. It is typically measured in a clinical setting using Cardiopulmonary Exercise Testing (CPET), where patients are evaluated while exercising to maximal capacity.
“We created an AI model, called TRUE-HF, trained on data from 154 patients and then validated on 63 patients, to estimate individuals' daily peak oxygen uptake using measurements from Apple Watch,” says Dr. Chris McIntosh, Senior Scientist at UHN and co-senior author of the study. “We found that when participants went about their daily routines while wearing an Apple Watch, our smartwatch-based pVO2 estimates strongly correlated with lab-derived ones from CPET.”
What’s more, they found that each 10% drop in the TRUE-HF–estimated fitness measure (pVO2) was linked to a more than threefold higher risk of an unplanned medical event. These events typically occurred approximately a week after the drop first appeared. A modified version of the model also predicted unplanned use of medical services around 21 days after the first drop in predicted pVO2. These results were further validated in a public cross-platform FitBit dataset from the National Institutes of Health (NIH) All of Us Research Program.
These results suggest that daily smartwatch measurements can provide early warning signs of worsening health and help predict when unplanned medical care may be needed for people living with heart failure. Identifying real-time changes in health through wearable technology, without requiring additional tests or added effort from patients, could enable faster, better care.
Yuan Gao is a doctoral candidate at UHN and co-first author of the study.
Dr. Yas Moayedi is a Clinician Investigator at UHN and co-first author of the study.
Dr. Christopher McIntosh, Senior Scientist at UHN and Dr. Heather Ross, Clinician Investigator at UHN, are co-senior authors of the study. Dr. McIntosh is an Assistant Professor in the Department of Medical Biophysics at the University of Toronto (U of T). Dr. Heather Ross is a Professor at the Institute of Medical Sciences at U of T.
Other study authors from UHN include Farid Foroutan, Bhavish Verma, Ben Kim, Enza De Luca, Margaret Brum, Darshan H. Bhrambhatt, Joe Duhamel, and Anne Simard.
This work was supported by the Ted Rogers Centre for Heart Research, the Natural Sciences and Engineering Research Council of Canada (NSERC), the Canadian Institutes of Health Research (CIHR), the University of Toronto, and UHN Foundation.
Dr. Ross is the Loretta Rogers Chair in Heart Function.
Apple Incorporated provided 200 iPhones and Apple Watch devices for the study, provided feedback on the manuscript, and collaborated with all authors to build the study-specific mobile application. All authors are investigating patenting the TRUE-HF model described in the manuscript.
Gao, Y, Moayedi, Y, Foroutan, F, Verma, B, Kim, B, De Luca, E, Brum, M, Brahmbhatt, DH, Duhamel, J, Simard, A, McIntosh, C, & Ross, HJ. Remote monitoring of heart failure exacerbations using a smartwatch. Nature Medicine. 2026 Mar 20. DOI: 10.1038/s41591-026-04247-3
×

Information Needs in Cancer Care
Patients and caregivers seek more inclusive, tailored resources for cancer care decisions.
Read MoreInformation Needs in Cancer Care
Patients and caregivers seek more inclusive, tailored resources for cancer care decisions.

Image Caption: Patients and caregivers are increasingly taking an active role in managing cancer care. To make informed decisions, they need clear, trustworthy, and accessible information. But many still face persistent challenges finding and using the health information they need.
Many people diagnosed with breast cancer rely on health information to understand their diagnosis and make decisions about their treatment. A new study from The Institute for Education Research at UHN shows that patients and caregivers often feel overwhelmed by the amount and complexity of health information and face persistent gaps in timely, inclusive, and tailored resources.
The research team interviewed 16 patients with breast cancer and caregivers to understand their experiences with accessing, evaluating, and using health information during diagnosis, treatment, and follow-up. Despite high education levels and digital literacy, many participants said they felt unprepared and overwhelmed by the volume and complexity of information presented to them. During the interviews, participants highlighted three main challenges:
● Mixed feelings about guidance from health care providers: Some participants felt reassured when clinicians cautioned against independent online research. Others, however, felt it limited their ability to actively participate in decision-making about their care.
● Information overload: Participants described difficulty navigating large amounts of information and finding resources that were timely, relevant, and appropriate for their specific stage of care.
● Need for more tailored and inclusive resources: Participants noted gaps in information, especially for male patients and people from racialized or cultural minority groups, who often could not find materials that reflected their experiences.
Participants emphasized the importance of connecting with other patients and caregivers. They also expressed strong support for a curated digital library that would bring trustworthy information together in one easy-to-access place.
These findings highlight opportunities for health care systems to strengthen information and resources available to support informed decision-making, promote patient participation, and create more equitable cancer care. Future research should explore the needs of patients who face additional barriers to accessing health information, including those with lower health literacy or limited support networks.
Mohamed Ugas, first author of the study, is a research analyst at the Princess Margaret Cancer Health Literacy Research Centre.
Dr. Janet Papadakos, senior author of the study, is a Scientist at The Institute for Education Research at UHN and the Co-Director of the Princess Margaret Cancer Health Literacy Research Centre. At the University of Toronto, Dr. Papadakos is an Assistant Professor at the Institute of Health Policy, Management, and Evaluation.
This work was supported by The Princess Margaret Cancer Foundation, with operational support provided by UHN Foundation.
Ugas M, Giannopoulos E, Tan J, Cil TD, Croke J, Forbes R, Giuliani ME, Koch A, Papadakos T, Quartey NK, Snow M, Westergard S, Papadakos J. Exploring the information needs of breast cancer patients and families in a large, urban, academic hospital: perceived barriers and facilitators to finding relevant and credible information. Support Care Cancer. 2026 Jan 23. doi: 10.1007/s00520-026-10344-3.
×
Research Institutes at UHN
Research conducted at UHN's research institutes spans the full spectrum of diseases and disciplines, including cancer, cardiovascular sciences, transplantation, neural and sensory sciences, musculoskeletal health, rehabilitation sciences, and community and population health.
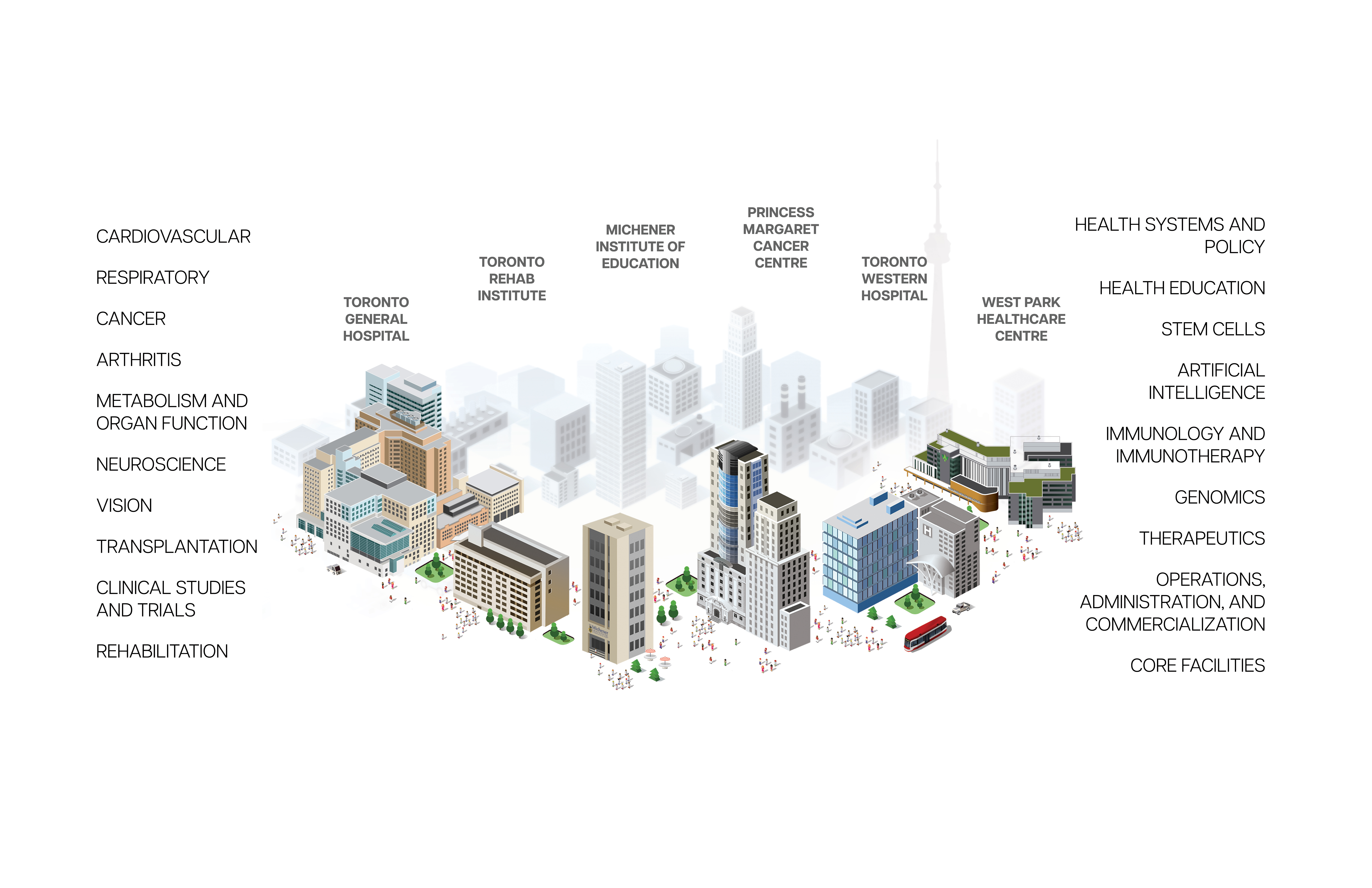
Research Institutes
Research at UHN is conducted under the umbrella of the following research institutes. Click below to learn more:





