The protein SETD7 is found in different types of cells and is implicated in many physiological processes including metabolism and inflammation. Researchers believe that SETD7 influences these processes by adding specific chemical tags known as methyl groups to other proteins. However, how this activity contributes to the normal functioning of cells is not fully understood by researchers, nor what role the disruption of SETD7 activity plays in disease.
A team lead by Dr. Cheryl Arrowsmith, a Senior Scientist at PM Cancer Centre, in partnership with Pfizer, has synthesized a new chemical compound intended for use by other researchers to shed further light on the biological role of SETD7.
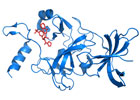
In collaboration with Pfizer, Dr. Arrowsmith’s team screened a library of 150,000 compounds and identified one that inhibited SETD7's activity. This “hit” compound was chemically modified to enhance its potency and facilitate its entry into cells. An atomic-level picture revealed that the modified compound, referred to as (R)-PFI-2, blocked SETD7 activity by attaching directly to it. Researchers then confirmed that (R)-PFI-2 could enter cells, bind tightly and specifically to SETD7 and, as a result, alter the activity of these cells.
“Our collaboration with Pfizer is very exciting. Pfizer is making (R)-PFI-2 freely available to researchers around the world to study the function of SETD7 and identify diseases that it might be able to help treat,” says Dr. Arrowsmith.
This work was supported by the Canadian Institutes of Health Research (CIHR), the Canada Foundation for Innovation (CFI), The Princess Margaret Cancer Foundation and the Structural Genomics Consortium which is a registered charity that receives funds from AbbVie, Boehringer Ingelheim, CFI, CIHR, Genome Canada, the Ontario Genomics Institute, GlaxoSmithKline, Janssen, Lilly Canada, the Novartis Research Foundation, the Ontario Ministry of Economic Development and Innovation, Pfizer, Takeda, and the Wellcome Trust.
(R)-PFI-2 is a potent and selective inhibitor of SETD7 methyltransferase activity in cells. Barsyte-Lovejoy D, Li F, Oudhoff MJ, Tatlock JH, Dong A, Zeng H, Wu H, Freeman SA, Schapira M, Senisterra GA, Kuznetsova E, Marcellus R, Allali-Hassani A, Kennedy S, Lambert JP, Couzens AL, Aman A, Gingras AC, Al-Awar R, Fish PV, Gerstenberger BS, Roberts L, Benn CL, Grimley RL, Braam MJ, Rossi FM, Sudol M, Brown PJ, Bunnage ME, Owen DR, Zaph C, Vedadi M, Arrowsmith CH. Proceedings of the National Academy of Sciences USA. 2014 Aug 18. [Pubmed abstract]
Image caption: Researchers used a powerful technique known as X-ray crystallography to obtain three-dimensional images of (R)-PFI-2 (red) bound to SETD7 (blue), as shown above. (PDB: 4JLC)




Comments